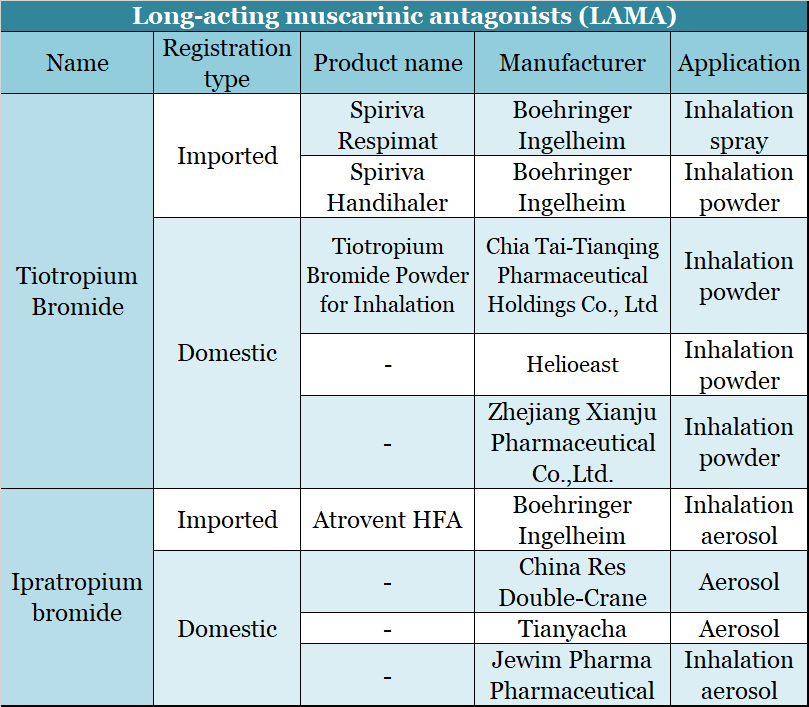 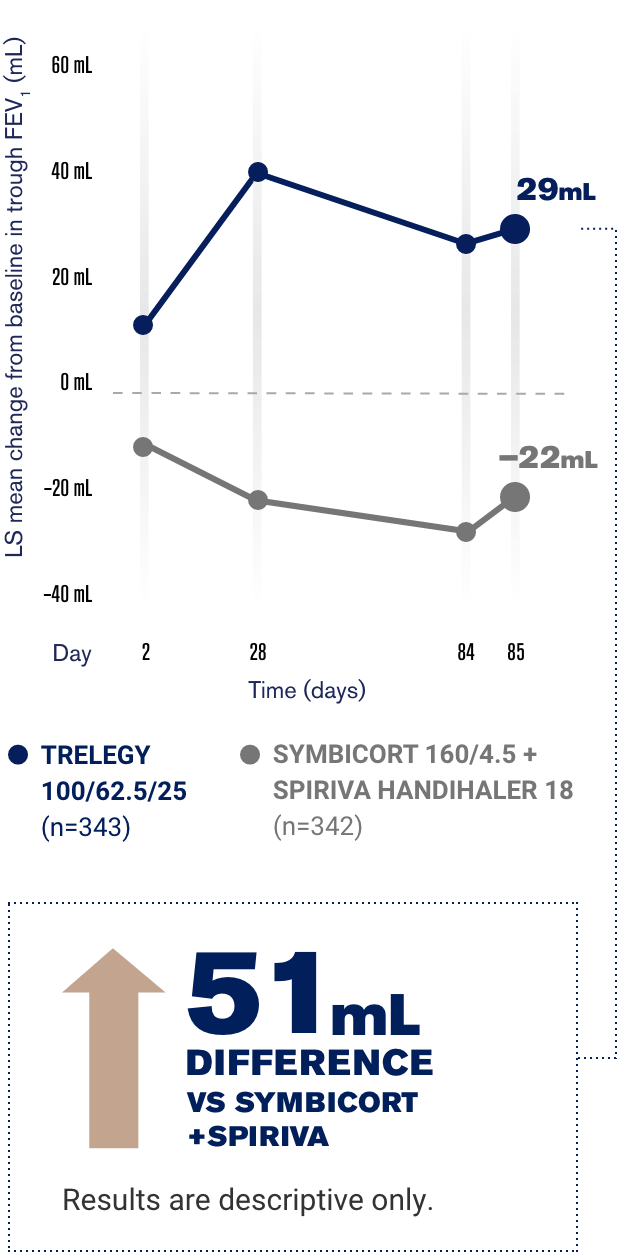
The Panel noted that the complainant had stated that the study cited in support of the claim was very specific whereas the claim was general. The Panel noted GlaxoSmithKline’s submission that FULFIL was the only trial referenced alongside the claim because it was the main study which referenced QoL as a primary endpoint. The Panel noted that the claim was referenced to the FULFIL clinical trial (Lipson et al 2017) which compared Trelegy Ellipta with AstraZeneca’s Symbicort Turbohaler (budesonide (ICS)/formoterol (LABA)). The Panel considered that the claim that Trelegy Ellipta demonstrated ‘Improvements in quality of life vs ICS/LABA’ unequivocally implied that the medicine improved quality of life in COPD patients compared with all ICS/LABA combinations. The detailed response from GlaxoSmithKline is given below. The complainant stated that, in essence, the claim was very general whereas the evidence base was very specific. The complainant noted that the claim ‘Improvement in quality of life vs ICS/LABA’ referenced the FULFIL clinical trial (Lipson et al 2017) which only compared Trelegy Ellipta with AstraZeneca’s Symbicort Turbohaler (budesonide (ICS)/formoterol (LABA)) no other ICS/LABA combination was assessed. This was followed by the two claims ‘The only COPD triple therapy delivered in a single daily inhalation’ and ‘Improvement in quality of life vs ICS/LABA’. In the centre of the screen was the headline ‘Less to take. The advertisement depicted the Trelegy inhaler in the bottom right hand corner of the screen. A complainant who described him/herself as a concerned UK health professional, complained about an advertisement (ref UK/TLY/0035/17K) for Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol) placed on the Primary Care Respiratory Society website by GlaxoSmithKline UK Limited in November 2018.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
